Omicron subvariant BA.2 reported in Canada: PHAC
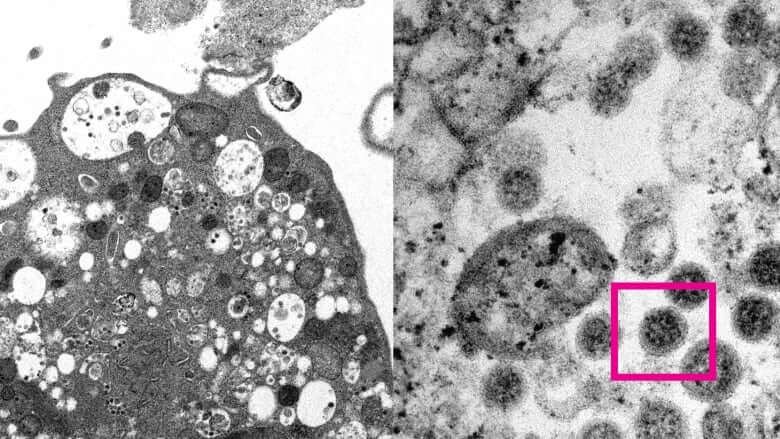
Unlocking the mysteries behind Omicron’s unique set of mutations will be key to understanding how this virus evolves and spreads — particularly as a tougher-to-detect subvariant is taking off in various countries, threatening to prolong this wave of infections.
The subvariant is known as BA.2 in the classification system used to catalogue the evolution of SARS-CoV-2, and it’s just one of a handful of Omicron subvariants featuring different mutations, which also includes the original lineage, as well as BA.1, BA.1.1, and BA.3.
BA.2 drew the close attention of virologists once it started to tick upward in multiple countries, including early signals of a slight rise here in Canada, suggesting it may be even more transmissible than its predecessor.
“There is a competition,” said Nathalie Grandvaux, a researcher and professor in the department of biochemistry and molecular medicine at the Université de Montréal. “It looks a bit like Delta versus Alpha … does that mean we will have another wave? It’s not something we can say for now.”
Denmark in particular is reporting a surge of BA.2 infections, with the subvariant accounting for nearly half of all Omicron cases by the second week of January, up from roughly 20 per cent over the end of 2021.
“During the same period, the relative frequency of BA.1 has dropped,” the Statens Serum Institut (SSI), a Danish research agency, reported on Jan. 20.
The U.K. is also monitoring BA.2 closely, and officials noted in a mid-January technical briefing that this subvariant is often able to fly under the radar. While BA.1 lacks one of the three target genes used in widespread SARS-CoV-2 testing, making it easy to spot — a process known as S-gene target failure — BA.2 can’t be detected the same way.
The same report noted that by Jan. 1, BA.2 accounted for five per cent of the U.K.’s S-gene positive tests — and that figure keeps rising.
That means the quick S-gene failure method to catch Omicron infections “is no longer sufficient to assess the spread of Omicron as a whole,” it continued.
By contrast, for much of the pandemic’s first year, it was far easier to track and manage this virus’s evolution.
That first changed when the B117 variant — later dubbed Alpha by the World Health Organization — burst onto the scene in late 2020, driving up case counts thanks to a set of mutations that helped it spread rapidly around the world.
As case counts ballooned, that evolutionary process has seemingly gone into overdrive, prompting variant after variant.
Strikingly, the differences between BA.1 and BA.2 are even greater than the differences between the original strain of SARS-CoV-2 and the Alpha variant, noted Denmark’s SSI.
On Monday, the World Health Organization said research into how BA.2 operates should be “prioritized independently” from other Omicron subvariants.
There are global signals the BA.2 subvariant is somewhat more transmissible, echoing the early days of Omicron’s spread, said Jesse Shapiro, an associate professor of microbiology and immunology at McGill University.
While it’s possible it may also drag on this wave of infections, he said, “there’s no big alarm bells so far.”

Questions over immune evasion
Yet questions remain about BA.2’s ability to reinfect a rising number of people who’ve already dealt with an Omicron infection. Could its dramatic evolutionary shift allow it to evade weeks-old immunity for the millions infected since late 2021?
There are a few documented instances of reinfections in Norway, so it’s possible BA.2 could infect some of those who’ve already recovered from BA.1 and push up case counts, said Anders Fomsgaard, chief physician and virus researcher at SSI, during a recent interview with a Danish media outlet.
But Matthew Miller, an associate professor of infectious diseases and immunology at McMaster, expects those kinds of reinfections are unlikely. What’s more worrisome, he suggested, are the impacts of a “little sister” to Omicron spreading among unprotected populations.
“People who’ve had only two vaccine doses, or no vaccine doses, or [are] incompletely vaccinated, they may be equally susceptible to BA.2 as they were to Omicron,” he said.
What’s reassuring about BA.2 — at least for now — is that the SSI’s initial analysis shows no difference in hospitalizations when compared to BA.1. Further research is underway to understand the subvariant’s impact on vaccine effectiveness, the SSI noted, but vaccines are expected to continue offering protection against severe illness.
The subvariant also comprises only a small percentage of Canada’s total number of known COVID-19 cases, federal data shows, though it does seem to be rising.
Omicron infections make up more than 90 per cent of Canada’s recent cases — and nearly all of those remain BA.1. But BA.2 infections totalled around one per cent of reported cases in early January, up from a mere 0.2 per cent in late December, with data for that stretch still accumulating.
Angela Rasmussen, a virologist with the Vaccine and Infectious Disease Organization at the University of Saskatchewan, stressed that those small numbers might not change, unlike the situation in Denmark, where BA.2’s potential takeover is cause for concern.
“What is important to remember here is that Denmark is not Canada. Denmark is a different population, with different vaccine uptake, different medical histories and demographics,” she said. “So what’s happening in Denmark isn’t necessarily going to automatically translate to Canada.”
Surveillance efforts will be crucial going forward to catch BA.2 cases and get a handle on its spread, Grandvaux noted, but she said that’s likely been a trickier task given its stealth nature.
“We need to keep the sequencing going on,” she said. “We need to know the behaviour of this virus.”
Scientists puzzled by Omicron’s origins
Darren Martin, a virologist at the University of Cape Town in South Africa, the country which alerted the world to Omicron’s existence, also said it’s crucial to understand why and how this variant evolved.
“We need to get serious, find out fast the conditions under which Omicron originated, the mechanisms at play during the assembly of its complement of mutations and figure out how we can minimize the probability of these conditions recurring,” he said in an email interview with CBC News.
“The last thing any of us want is another new hyper-evolved [variant of concern] cropping up every few months.”
Martin and a team of researchers recently shared early findings regarding the original Omicron lineage, noting more than a dozen of BA.1’s high number of mutations are highly rare, suggesting they wouldn’t necessarily be helpful to an evolving virus.
Instead, those unique quirks are part of a constellation of mutations which appear to grant Omicron the uncanny ability to spread rapidly between human hosts, leaving other variants in its dust.
What’s concerning, Martin said, isn’t just that puzzling original Omicron lineage, but the fact that BA.2 and other lineages exist as well — all closely related, yet with unique properties, raising questions over how each evolved fast enough to be discovered by scientists around the same time.
“How did a virus with a much lower mutation rate than viruses like HIV and influenza manage, in a linear, stepwise, process, to string together such large sets of interacting mutations, when each mutation on its own would have reduced its fitness?” Martin questioned.
“The conditions under which this evolution occurred might need to be quite different than those of a normal short-term SARS-CoV-2 infection.”
Some scientists have theorized that Omicron developed during a long-lasting infection in an immunocompromised patient, or within animal hosts before suddenly spreading back to humans with explosive force.
However Omicron came to be, its existence — and continual shape-shifting — hints at this virus’s capacity for radical evolution if infections remain at high levels around the world.
“It behaves like we could predict,” Grandvaux said. “It mutates as it’s transmitting.”








Redes Sociais - Comentários